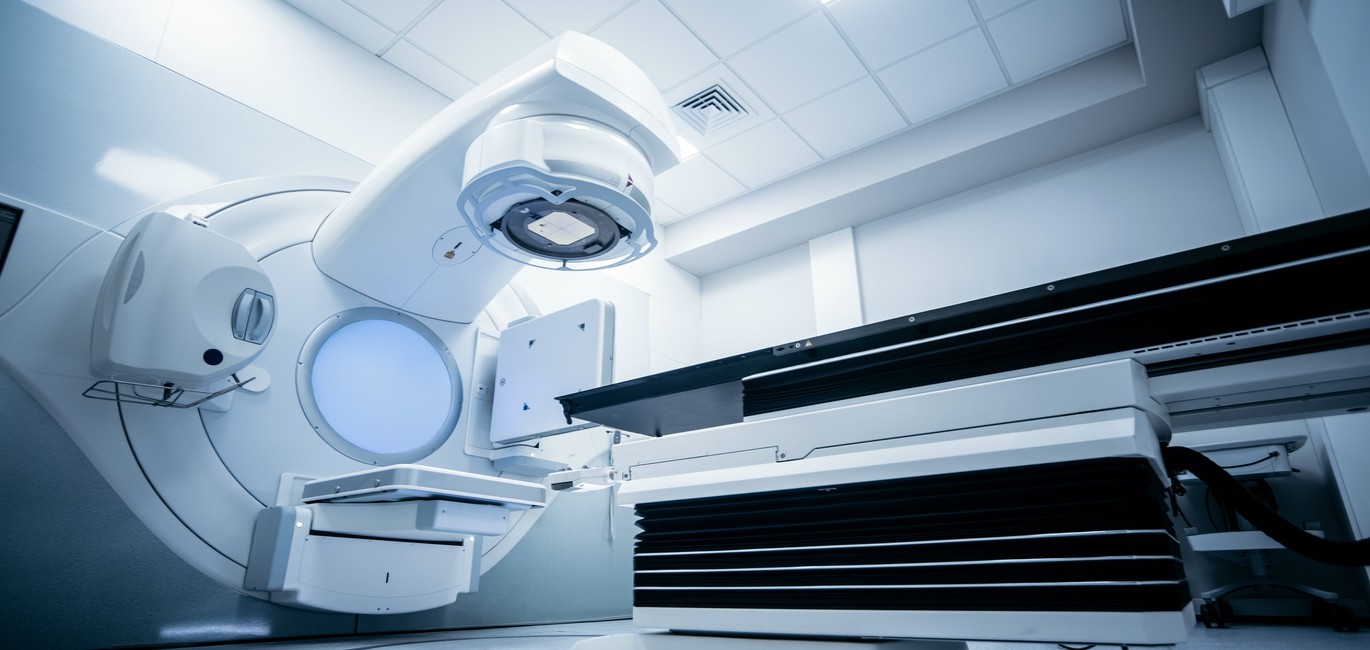
Overview
Pharmaceuticals, Nutraceuticals and Medical Devices sit at the core of MCI’s business, with pharmaceuticals representing the largest and most established industry segment of our portfolio.
Underpinned by the security of Lloyd’s and decades of life science market experience, MCI brings both extensive sector knowledge (through in house ex-industry and academic individuals) and commercial awareness to all our underwriting decisions. Our cross-class underwriting expertise also enables a broad, integrated approach to risk assessment, facilitating our offering.
This combination of technical underwriting strength, market insight and financial reliability positions MCI as a long-term partner to clients operating in what is a highly regulated and commercially demanding environment.
Target sectors & Company types
Clinical Trials:
- Phase I, II, III and IV trials
- Bio-equivalence and investigator-led studies
- Sponsored trials of drug, medical device, nutraceutical and biotechnology companies
- Contingent exposures of hospitals
- Charities and educational establishments
- Biotechnology associations
Pharmaceuticals:
- Patented Molecules
- Generic drugs (off patent)
- Final Formulation medicines
- Over the counter products
- Pharmaceutical intermediates/catalysts
- Fine Chemicals
- Blood fractionation/filtration service providers/blood banks
Nutraceutical & Dietary Supplements:
- Vitamins and minerals
- Herbs and Botanicals
- Homeopathic remedies
- Topical Skincare products
- Protein Supplements
- Weight gain supplements
- Weight loss supplements
- Sexual enhancement products
- Energy Products
- Pre/Post-natal products
Medical Devices:
- Hospital Consumables, I.e. laryngoscopes, syringes, scalpels
- Temporarily invasive medical equipment, I.e. urinary catheters, IV drips
- Breast implants
- Orthopaedic screws
- Dental implants
- Diagnostic devices (X-Rays, CT scanners, MRIs, pulse oximeters)
- Wound dressings
- Stents and other cardiac products
- Permanently invasive devices (narrow appetite here)
Life Science Service-oriented Businesses:
- Clinical Research Organisations (CROs)
- Contract Research Services (CRS)
- Medical Writing
- Regulatory Consultants
- Biostatistics organisations
- Testing Companies
- Medical testing for patients where no interpretation of results is given (eg. genetic testing or blood sample testing)
- Contamination testing for batch release of Nutraceuticals and Pharmaceuticals
- Cleanroom commissioning
COMPANY TYPES
- Manufacturers of own products
- Contract Manufacturers
- Packagers and Labellers
- Wholesalers and distributors
- Ingredient importers, distributors and manufacturers
- Retail Stores (of dietary supplements)
- Product licence holders
- Biotechnology Companies
- Research and Development
- Custom machining risks (Medical Devices)
COMPANY SIZE
- All sizes of business considered; with
- Nil revenue start-up companies to USD500m of USA derived revenues actively targeted
- >USD500m of USA derived revenue considered on a case-by-case basis.

Coverage & Line Size
Line Size
- USA (parented entities) – USD10M Maximum line size, any one claim and in the aggregate.
- Worldwide – USD 20M Maximum line size, any one claim and in the aggregate.
Deductible Structure
- Minimum deductible is USD2,500 each and every claim
XS or Primary Capacity
- MCI prefers leading primary placements, though will write excess placements subject to underlying wording review
COVERAGE
- Claims Made Products; but
- Occurrence Products cover possible for low hazard products and certain medical devices
- General Liability / Premises Liability / Public Liability – claims made or occurrence
- Professional Indemnity / Errors and Omissions (“E&O”) / Manufacturers E&O – Claims Made basis
- Clinical Trials – Legal liability or No Fault basis / Trial duration or Annually renewable
Standard Endorsements & Extensions
- Drug & specified dietary supplement / medical device exclusion list
(NB : This is not a mandatory list for us and can be adapted to suit individual client needs, subject to satisfactory information)
- Employers Liability
- Pollution liability (including site specific & gradual via our EIL team)
- Excess Auto and contingent / non-owned Auto
- Product Recall
Why we’re different
- Extensive Life Science Expertise - Covering pharmaceuticals, nutraceuticals, and medical devices across R&D, manufacturing, and distribution.
- Broad Underwriting Appetite - Continuously monitoring the market to provide coverage for hard-to-place and emerging Life Science risks.
- Trusted & Experienced - Decades of market knowledge and strong financial capacity provide stability in complex business environments.
- Tailored Coverage - Flexible, client-focused solutions for complex exposures.



